 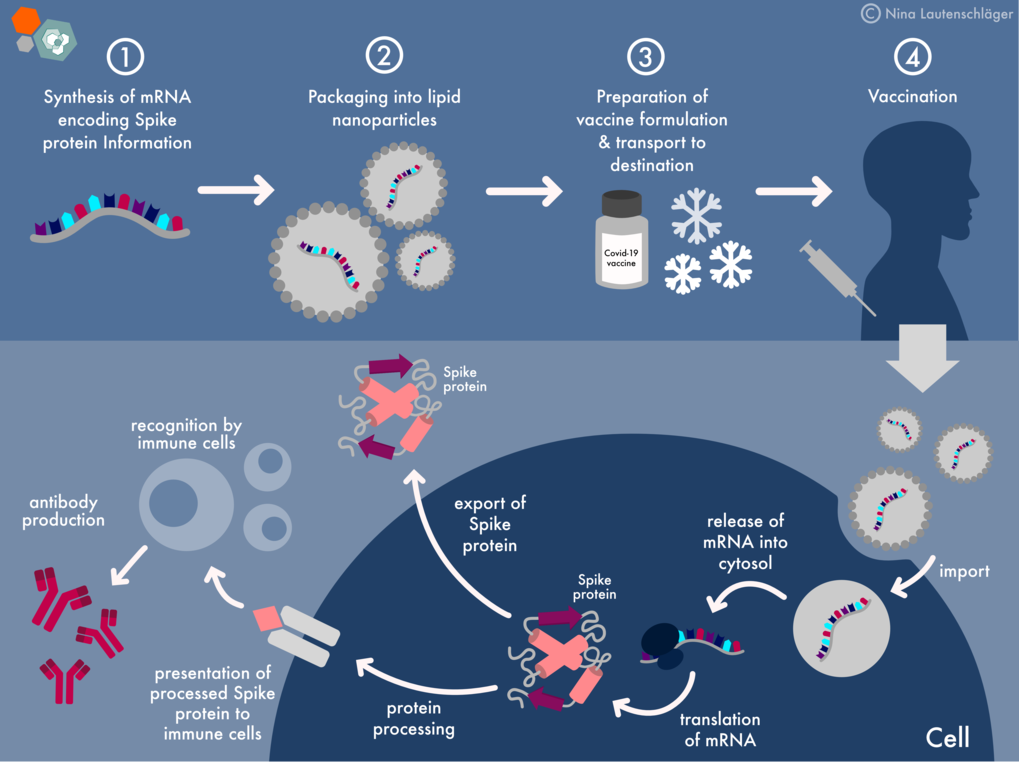 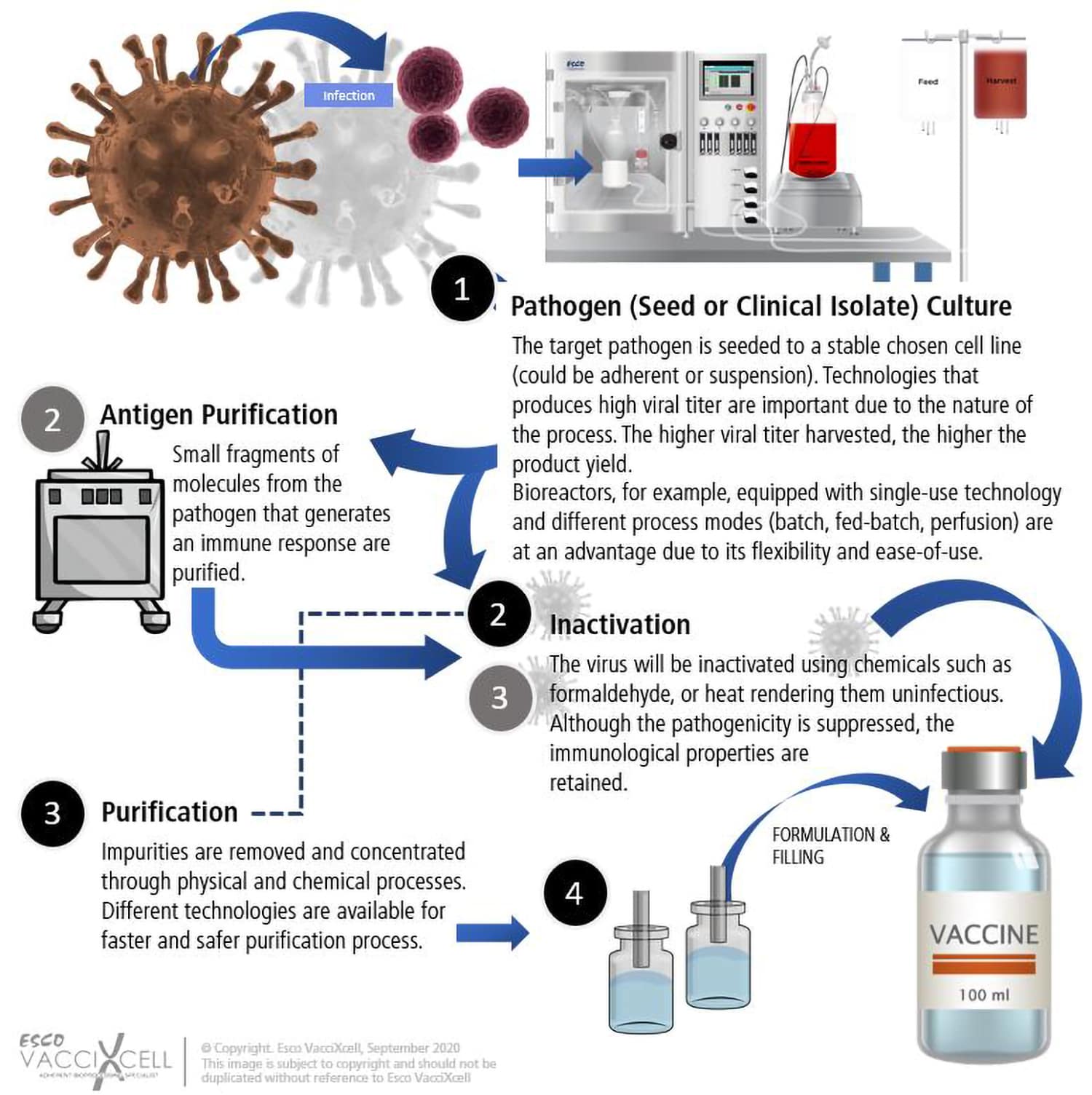
How COVID has shaken up the industry landscape That would make for a faster rate of growth among these third-party players than in the pharma industry itself. A report from Grand View Research finds that the global contract-manufacturing and development market for drugs will balloon to $157.7 billion by 2025, representing a 6.9% compound annual growth rate over seven years. Biopharmaceutical firms’ reliance on outsourced manufacturing for drugs of all stripes is a growing market, according to analysts. And producing that initial substance, the “bulk” biological substance that makes up the core of something like a vaccine, is no mean feat.īut despite the less than ideal business conditions, this type of manufacturing is only gaining steam. The ever-changing innovation in the field requires these manufacturers to have sharp in-house scientific expertise and the willingness to take risks on unproven products during an emergency. Dozens upon dozens of other coronavirus vaccines are in various stages of clinical trials or authorization around the world, encompassing pharmaceutical giants and life science industry upstarts alike.īut once a product is proven, many pharma companies rely on outsourced manufacturing. Johnson & Johnson and AstraZeneca both utilize something called an adenovirus, or cold virus, to create their vaccines, although J&J uses a human adenovirus while AstraZeneca uses one from a chimpanzee. These include companies like Pfizer and its partner BioNTech, and Moderna, which both leveraged pioneering mRNA technology to win the first authorizations for COVID vaccines in the U.S. It might be easy to think that drugmakers, the so-called innovator companies behind a pharmaceutical product, are the only real parties involved in rolling out a new therapy. What are these companies and what do they do, anyway? What happens for individual contract-manufacturing firms going forward is a far more nuanced story. 
More than 192 million doses have already been administered across the country as of April 13, according to the CDC. There are just three emergency-authorized COVID vaccines in the U.S., with Johnson & Johnson’s on a temporary pause over potential safety concerns. During a public health emergency, ad hoc relationships must be and have been struck between rivals and industry players globally to ensure that as many vaccine doses as feasible wind up in as many arms as possible, industry executives relayed to Fortune. Middlemen abound in the life sciences business generally. “So then we see that the hardware, the volume of manufacturing, that is available for worldwide production meets the demand.” “The good news is that there is enough capacity worldwide,” says Simone Blayer, a chemical engineer and pharma industry veteran who is now the head of chemistry, manufacturing, and control at the nonprofit PATH, which works to help improve vaccine production and equity around the world. And still others may have to take on bigger roles in manufacturing non-COVID pharmaceuticals as giants like Pfizer focus their own manufacturing facilities on vaccine production. Some of the giants in the drug contract-manufacturing space may benefit from knowledge of a certain coronavirus vaccine technology to maximize doses others may not have the capacity or know-how to create customized biological ingredients relying on new vaccine platforms such as Pfizer’s and Moderna’s messenger RNA (mRNA)-based tech, but still may be able to scale up other kinds of COVID shots from companies like Johnson & Johnson or AstraZeneca. The COVID outbreak has led to a spider’s web of relationships among these third-party contract firms and the pharma industry partners they work with, and experts tell Fortune that it’s tough to predict outright how the pandemic may reshape these outsourcing companies’ bottom lines and their business strategies-not to mention what their roles will be in the future as variant strains of the coronavirus emerge. These are the companies that help manufacture drug doses or carry out other important functions that not all drugmakers have the capacity to do, such as putting a COVID vaccine into vials so they can actually get to patients. That makes on-the-ground contract manufacturers-companies that may not be household names but serve a critical role in the drugmaking process-particularly important in this moment. And then you have to actually make this new product at scale so that shots can be delivered to vaccine sites and administered into arms. Simply inventing a new vaccine or therapeutic is a time-consuming and unpredictable process in and of itself. When you have a global pandemic like COVID brushing up against the labyrinthine business dynamics of the drug industry, things can get tricky. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
